CryoLetters Volume 47 - Issue 4
CryoLetters 47 (4), 235-248 (2026)
© CryoLetters, editor@cryoletters.org
doi.org/10.54680/fr26410110112
PERSPECTIVE: The role of encapsulation in the development of plant cryopreservation techniques
María Teresa González Arnao1*, Florent Engelmann2 and Takao Niino3
- Facultad de Ciencias Químicas, Universidad Veracruzana, Orizaba, Veracruz, Mexico.
- IRD, 911 avenue Agropolis, BP 64501, F-34394 Montpellier Cedex 5, France.
- Formerly, Gene Research Center, University of Tsukuba, Tsukuba, Japan.
*Corresponding author’s E-mail: teregonzalez@uv.mx
Abstract
Over the past 35 years, encapsulation technologies have played an important role in the development and diversification of cryopreservation techniques for cells and organized structures from numerous plant species. Encapsulation with calcium alginate was initially developed as a technological approach for the production of synthetic seeds. However, the coating of biological material with a hydrogel matrix subsequently promoted new applications that provided additional benefits to in vitro multiplication and preservation techniques. Alginate coatings can be complemented with growth regulators, antioxidants, and nanoparticles. These compounds have helped mitigate the toxic effects of cryoprotectants by regulating their penetration rate and have enabled the implementation of drastic desiccation treatments that would have otherwise been lethal. Furthermore, encapsulation facilitates the simultaneous handling of large quantities of samples, simplifying the ongoing handling required. Cryopreservation protocols based on encapsulation include Encapsulation-Dehydration, Encapsulation-Vitrification, V- and D-cryoplate methods. The objective of this review is to provide information on the impact of encapsulation of plant material on the advancement of cryogenic procedures and, consequently, on the understanding of tolerance of plant tissues to cryopreservation. In view of the advantages of encapsulation, it is considered that it will facilitate the long-term conservation of a greater number of plant species.
Keywords: calcium alginate beads; cryoplates; growth recovery; liquid nitrogen; vitrification-based procedures.
CryoLetters 47 (4), 249-268 (2026)
© CryoLetters, editor@cryoletters.org
doi.org/10.54680/fr26410110312
PERSPECTIVE: Recent advances in sperm cryobiology: nanomaterial - assisted preservation, AI-based quality assessment and cellular protection strategies
C. Prasanth, A.S. Vickram*, S. Bharath, B. Bhavani Sowndharya and A. Saravanan
- Department of Biotechnology, Saveetha School of Engineering, SIMATS, Chennai, 602105, India.
*Corresponding author’s E-mail: vickramas.sse@saveetha.com
Abstract
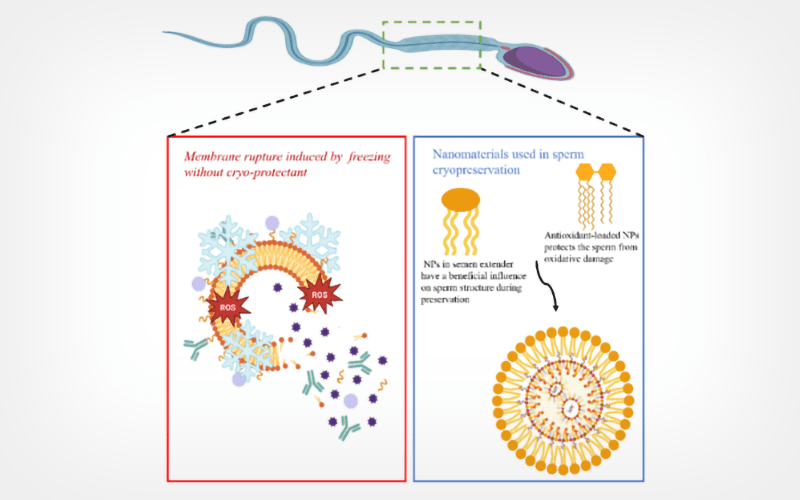
Modern biological and medical technologies require low-temperature storage of cells, tissues, and tissue-engineered constructs to facilitate their transfer and to prolong their usability prior to application. Many industries, including biological research, medicine, and agriculture, depends on low-temperature preservation to preserve the integrity and viability of cells, tissues, and organs for long periods of time. Nanotechnology has had an influence on low-temperature preservation in recent years, providing advanced solutions that greatly enhance biological sample storage. Development of metal oxide and inorganic metal nanoparticles (MNPs) has drawn interest in several biotechnological and medicinal domains, including cryopreservation. The integration of MNPs into reproductive biology protocols represents a novel and emerging research area. Although AI has long been used in medicine, the recent rise of deep learning is a major factor in the growth of this field. Sperm morphology categorization has been automated using deep learning frameworks that focused on different sperm cell components. ROS generation is significantly elevated during sperm cryopreservation. Low, medium, and high ROS levels in cells cause apoptosis, auto-phagocytosis, and necrosis, respectively. Antioxidants can be added to semen extenders to reduce the elevated ROS levels during cryopreservation.
Keywords: antioxidants; artificial intelligence; automated cryopreservation; deep learning; metal oxide nanoparticles; nanomaterials.
CryoLetters 47 (4), 269-286 (2026)
© CryoLetters, editor@cryoletters.org
doi.org/10.54680/fr26410110612
Evaluation of cryopreservation strategies in sugarcane true seeds using Kaplan-Meier survival analysis
C. Jayabose*, V. Anusheela, A. Annadurai, M. Prabakaran, R. Valarmathi and D. Neelamathi
- Division of Crop improvement, Indian Council of Agricultural Research, Sugarcane Breeding Institute, Coimbatore–641 007, Tamil Nadu, India.
*Corresponding authors’ E-mail: cjayabose@gmail.com
Abstract
Background
Cryopreservation offers a robust approach for the long-term conservation of sugarcane (Saccharum spp.) germplasm, particularly for maintaining the viability of true seeds used in hybridization programs. According to conservation biology, any plant part capable of regenerating a whole plant can be conserved. However, the effectiveness of cryogenic treatments in preserving sugarcane true seeds requires systematic evaluation.
Objective
To evaluate the survival dynamics of five sugarcane genotypes under different cryogenic treatments and to determine the efficacy of silica gel dehydration combined with liquid nitrogen exposure in enhancing cryo-tolerance.
Materials and methods
Five sugarcane genotypes (Co 1148, 1148-13-11-2-255, CoSe 92423, CoM 9217, and LG 07590) were subjected to four storage treatments: (i) silica gel dehydration followed by liquid nitrogen (Si+LN+), (ii) direct liquid nitrogen storage without dehydration (Si−LN+), (iii) low-temperature storage at −23 °C, and (iv) non-cryogenic control. Survival probabilities over 21 days were analyzed using the Kaplan–Meier method with NCSS software. Survival functions were compared across treatments using logrank tests.
Results
Across genotypes, Si+LN+ consistently showed the highest survival probabilities over a short-term post-storage assessment duration. Survival curves under Si+LN+ significantly differed from controls and low-temperature treatments (p < 0.001). Among the genotypes, Co 1148 and 1148-13-11-2-255 exhibited the greatest cryo-tolerance, maintaining survival >40% after 21 days, whereas CoM 9217 was the most sensitive. Incorporation of silica gel dehydration effectively mitigated cryo-injury during liquid nitrogen exposure.
Conclusion
Kaplan–Meier survival analysis provided detailed temporal insights into treatment–genotype interactions, offering more robust statistical evaluation than endpoint germination alone. The findings emphasize the utility of silica gel dehydration in enhancing cryogenic survival and highlight the Si+LN+ protocol as optimal for conserving sugarcane reproductive viability in germplasm conservation programs. The controls maintained higher survival only over the short experimental timeframe, whereas cryopreserved seeds are intended for indefinite storage without genetic or physiological deterioration.
Keywords: cryopreservation; germplasm conservation; Kaplan–Meier analysis; sugarcane; silica gel dehydration; true seeds.
CryoLetters 47 (4), 287-296 (2026)
© CryoLetters, editor@cryoletters.org
doi.org/10.54680/fr26410110712
The effect of carboxymethyl cellulose on gene expression related to apoptosis, cold shock, motility, and reactive oxygen species of cryopreserved dog sperm
Saddah Ibrahim1,2#, Joonho Moon1#, Mohamed Abdou1,3 and Il-Jeoung Yu1*
- Laboratory of Therigenology and Reproductive Biotechnology, College of Veterinary Medicine, Jeonbuk National University, and Biosafety Research Institute, Iksan 54596, Republic of Korea.
- Department of Veterinary Medicine and Surgery, College of Veterinary Medicine, Sudan University of Science and Technology, P.O. Box 204, Hilat Kuku, Khartoum North 11111, Sudan.
- Department of Theriogenology, Faculty of Veterinary Medicine, Assiut University, Assiut, Egypt.
*Corresponding author’s E-mail: iyu@jbnu.ac.kr
#Contributed equally as first authors
Abstract
Background
Carboxymethyl cellulose (CMC), a cellulose derivative, is osmotically inert and has low viscosity compared with other non-permeating cryoprotectants.
Objective
To assess whether a combination of CMC with glycerol can improve the efficiency of freezing extender for dog sperm cryopreservation.
Materials and methods
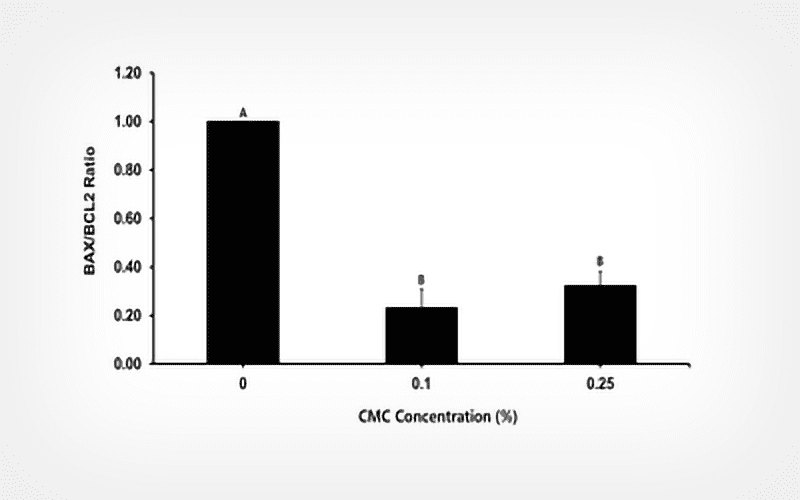
Dog spermatozoa were diluted and frozen in freezing extender supplemented with 0% (control), 0.1%, 0.25%, 0.5%, or 0.75% (w/v) CMC. Post-thaw, the sperm kinematic parameters (CASA), plasma membrane integrity (SYBR-14/PI), and acrosome integrity (FITC-PSA) were evaluated. The expression of genes related to apoptosis (BCL2, BAX, and ANAXA), motility (BACTIN), cold shock proteins (YBX3), and ROS (CAT, ROMO1, and SMOX) was evaluated using qPCR.
Results
The total motility was higher in the 0.1 % CMC group compared to the other groups (P < 0.05). In addition, the expression of the antiapoptotic gene BCL2 was upregulated in the 0.1% CMC group compared to the control, whereas the gene expression of BAX and SMOX exhibited lower levels in the 0.1% and 0.25 % CMC groups than the control group (P < 0.05). The expression of YBX3 genes was significantly decreased with 0.25 % CMC supplementation.
Conclusion
The addition of CMC to a freezing extender improved sperm motility and alleviated cryostress by regulating the gene expression of BCL2, BAX, SMOX, and YBX3.
Keywords: carboxymethyl cellulose; cryopreservation; dog sperm; gene expression.
CryoLetters 47 (4), 297-309 (2026)
© CryoLetters, editor@cryoletters.org
doi.org/10.54680/fr26410110212
Effects of developmental age on cryopreservation and cold storage of seeds of Arundina graminifolia, a classic Dai medicinal orchid
Huan Dai1,3, Kanchit Thammasiri1, Lei Fan2, Junchao Ma2, Qian Xu2, Fuchuan Wu1, Hugh W. Pritchard2 and Xiaojian Hu2*
- Xishuangbanna Tropical Botanical Garden, Chinese Academy of Sciences, Menglun, Mengla, Yunnan 666303, China.
- The Germplasm Bank of Wild Species, Kunming Institute of Botany, Chinese Academy of Sciences, Kunming, Yunnan 650201, China.
- University of Chinese Academy of Sciences, Beijing, China.
*Corresponding author’s E-mail: huxiaojian@mail.kib.ac.cn
Abstract
Background
Arundina graminifolia is a traditional medicinal plant of the Dai people in China. To enhance its ex situ conservation greater knowledge is needed on the species’ seed storage behavior, particularly the effects of developmental age, moisture content and temperature and storage time on seed survival (germination).
Objective
To investigate whether A. graminifolia seeds can be preserved using conventional seed banking and/or cryopreservation in relation to seed developmental age and lipid thermal fingerprinting.
Materials and methods
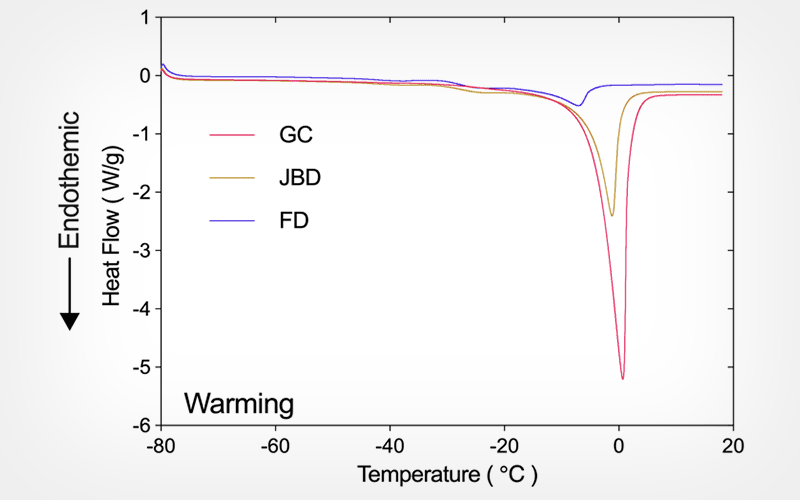
Naturally pollinated and artificially pollinated capsules at three maturity stages were collected (i.e., six seed lots), equilibrated to 15%-75% relative humidity (RH), and stored at 4℃, -20℃ and in liquid nitrogen (LN). Seed quality, including after 3 and 6 months of storage, were evaluated using asymbiotic germination. Seed water and lipid thermal properties were determined by differential scanning calorimetry.
Results
Seed maturity state was confirmed by the moisture content decrease of fresh seeds as well as the lipid transition enthalpy increase (from c. 10 to 20 J g-1 DW) and melting peak temperatures of dry seeds. Immature seeds (either as green capsules [GC] or 60 days after pollination [DAP60]) had some level of desiccation sensitivity (up to germination halving) and significant losses of germination during storage. Mature seeds (from capsules just before dehiscence [JBD], fully dehisced [FD], DAP90 and DAP120]) retained high germination levels under the vast majority of RH x Temperature x Time combinations. DSC analysis of the enthalpy of transitions during cooling and warming revealed that seeds equilibrated to ≤ 75% RH were below the unfrozen moisture content.
Conclusion
Mature seeds of A. graminifolia seem to have orthodox seed storage behaviour, and both cold storage and cryopreservation appear feasible for the long-term ex situ conservation of this species.
Keywords: Arundina graminifolia; cryopreservation; differential scanning calorimetry; maturity; Orchidaceae; seed storage behaviour.